ImmunityBio, Inc.
@ImmunityBio
Followers
2,593
Following
114
Media
239
Statuses
583
ImmunityBio is developing cell and immunotherapy products that are designed to help strengthen each patient’s natural immune system.
Culver City, CA
Joined March 2020
Don't wanna be here?
Send us removal request.
Explore trending content on Musk Viewer
オーロラ
• 492250 Tweets
Joost
• 334472 Tweets
#الهلال_الحزم
• 85607 Tweets
Fulham
• 81859 Tweets
#SixTONESANN
• 78237 Tweets
Man City
• 62845 Tweets
自分これ
• 61859 Tweets
Gvardiol
• 54232 Tweets
Ali Koç
• 47760 Tweets
Dremo
• 41197 Tweets
太陽フレアのせい
• 39467 Tweets
Burnley
• 33746 Tweets
SEE YOU NEXT MONTH JIN
• 31685 Tweets
دوري روشن
• 30597 Tweets
الدوري الاقوي
• 28769 Tweets
Granada
• 25210 Tweets
روشن السعودي
• 24591 Tweets
Luton
• 22315 Tweets
Haley
• 22196 Tweets
やまとなでしこ
• 14763 Tweets
الدوري التاريخي
• 14541 Tweets
#الهلال_بطل_الدوري_التاريخي
• 11927 Tweets
Last Seen Profiles
Thrilled about the news that our partners Drs. Hotez and Bottazzi have been nominated for the Nobel Prize for their work on low-cost
#COVID
-19 vaccine that’s already in use in India. Congratulations on the well-deserved recognition!
@TexasChildrens
@PeterHotez
@mebottazzi
3
37
271
This week's
@US_FDA
approval of a new treatment option for BCG-unresponsive non-muscle invasive bladder cancer has the potential to represent a new standard of care for patients. Learn more:
#BladderCancer
$IBRX
5
31
88
In an interview today with
@YahooFinance
, ImmunityBio Chairman,
@DrPatSoonShiong
, spoke with
@AnjKhem
about what the approval of
#Anktiva
means for ImmunityBio as well as investors. Check out their conversation here ⬇️ $IBRX
4
22
88
ImmunityBio and
@MSFTResearch
donate a combined 24 petaflops of GPU computing power to create models for researchers working on
#coronavirus
vaccines & therapeutics. Models can be derived in days, rather than the months it would normally require.
4
32
77
“Until we have a second-generation vaccine that actually stops transmission, [the pandemic] will not be over” shared ImmunityBio Founder
@DrPatSoonShiong
today on
@QuestMeans
. ImmunityBio is proud to begin phase 3 clinical trials for our COVID vaccine in Africa next week.
$IBRX
11
30
73
Tomorrow, we are participating in the
@WhiteHouse
summit on next-generation
#COVID
vaccines. Our vaccine candidates are designed to thwart future coronavirus variants and provide longer duration protection. Tune in here: .
@WHCOVIDResponse
8
18
64
We're pleased to share the FDA has accepted for review our BLA for N-803+BCG for BCG-unresponsive NMIBC. With this important step, we're closer to being able to offer a promising combo therapeutic to people living w/
#NMIBC
and, ultimately, reducing the incidence of cystectomies.
3
9
59
Together with
@AAHI
&
@Amyris
, we've embarked on a path to develop 2nd gen RNA vaccines for
#COVID
that provide broad, durable protection. A better future exists if the federal government acts to financially and scientifically support 2nd generation vaccines.
@WHCOVIDResponse
2
11
50
We’re excited to partner with the Infectious Disease Research Institute (IDRI) and
@Amyris
on a second-generation Covid-19 RNA vaccine. This is sorely needed as the U.S. is averaging more than 300,000 new cases a day for the first time in the pandemic.
2
16
50
Reached new milestone with our
#BladderCancer
trial: > 80 subjects in the study have completed at least 12 months of follow up & results continue to demonstrate clinically meaningful, sustained benefit. We expect to submit BLA to the FDA in April. $IBRX
11
10
51
This USA TODAY article provides an overview of second-generation COVID-19 vaccines; ImmunityBio is one of a few vaccine developers studying multiple delivery routes.
Learn more:
#USATODAY
#SecondGenerationCOVID19Vaccine
#COVID19Vaccine
$IBRX
11
15
45
We’re honored to partner with IDRI and Baylor College of Medicine to expand our COVID-19 vaccine program. These 2nd gen vaccines are designed to confer long-term immune memory and are easier to make, store and distribute.
$IBRX
@mebottazzi
@PeterHotez
2
16
48
"A variant-proof vaccine could slow the transmission of Covid-19, and stopping such transmission is the only way we can move past the pandemic," said Patrick Soon-Shiong, in an article for
@BBC
by
@DrDavidACox
about the future of
#Covid19
#vaccines
.
5
6
47
ImmunityBio is committed to developing novel treatments and vaccines for cancer and infectious disease, including COVID-19. Working with AAHI and other talented scientists, we want to change the current paradigm of care for these diseases.
#GlobalCOVIDSummit
@AAHITweets
18
12
47
ImmunityBio gains $300 million in non-dilutive capital for commercialization and pipeline expansion efforts.
To learn more, read the full press release here:
$IBRX
#CancerResearch
#immunotherapy
#BladderCancer
3
6
49
🚨 On the heels of the FDA approval of our combination therapy for treatment of
#NMIBC
, ImmunityBio and
@seruminstindia
have agreed to an exclusive arrangement for global supply of BCG. Read More:
3
24
97
"Technology is here. We have the solutions. We just have to implement them correctly," said Dr. Sandeep Bobby Reddy, Chief Medical Officer at ImmunityBio. What will next-gen vaccines mean? "Cross reactivity and memory from a vaccine will give us confidence."
@WHCOVIDResponse
3
14
46
In a discussion with
@UroCancerMD
for
@urotoday
,
@DrPatSoonShiong
discussed the effectiveness, safety and tolerability of N-803 for BCG unresponsive NMIBC. Read more here:
#BladderCancer
$IBRX
3
11
39
Join us at
#AUA24
for a groundbreaking discussion on next-gen immunotherapy for bladder cancer with
@UroCancerMD
and
@DrPatSoonShiong
. Details: Friday. 5/3 @ 3:15 CDT in the Learning Lab at AUA Square.
#NMIBC
#Immunotherapy
1
11
47
.
@DrPatSoonShiong
's life quest is to find a
#cancer
vaccine. He has promised to “connect the dots” of how to get there and the underlying basis of the
#CancerMoonshot
. The
@USUhealthsci
2023 David Packard Award Lecture begins to connect these dots:
$IBRX
12
11
41
#ChapelHill
@BCAN
#Bladdercancer
Walk: ImmunityBio is proud to co-sponsor the BCAN Walk to End Bladder Cancer. Thank you BCAN for the important work you do everyday on behalf of bladder cancer patients and their caregivers.
1
6
43
This excellent overview by Atlantic writer Katie Wu illustrates the important role T cells are playing in second-generation COVID-19 vaccines. Learn more:
#COVID19Vaccine
#ImmunityBio
#TheAtlantic
$IBRX
8
17
38
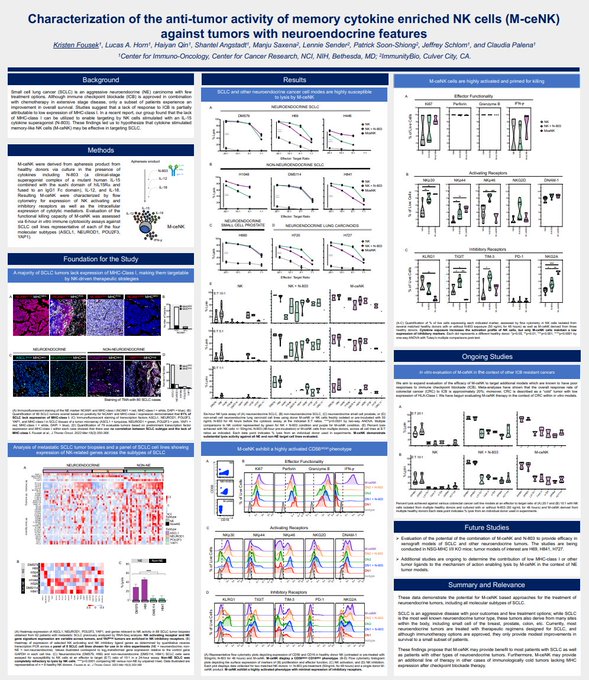
Exciting news from
#SITC23
! First data on our Memory Cytokine-Enriched NK Cells in Small Cell Lung Cancer reveals strong anti-tumor potential. 🦠🔬
Read more:
#CancerResearch
$IBRX
2
5
41
Today we announced results from our Phase 2 Metastatic Pancreatic Cancer trial at
#ASCOGI
, showing that our combination immunotherapy, Nant Cancer Vaccine, is potentially effective in pancreatic cancer where few treatment options exist.
$IBRX
5
12
37
In an interview with
@TeamCavuto
on
@FoxBusiness
, ImmunityBio founder,
@DrPatSoonShiong
, highlighted the unique mechanism of action for Anktiva and reiterated our belief in Anktiva and its potential efficacy as a next-generation immunotherapy across multiple tumor types. $IBRX
0
13
57
#ICYMI
: Our Chief Medical Officer, Bobby Reddy, M.D., recently spoke with
@BioProcessOL
@mattpillar
about N-803 and its potential for
#NMIBC
, and what it takes to bring a drug to market. Listen here:
9
5
39
Most cancer treatment plans weaken the immune system. At ImmunityBio, we work to create targeted immunotherapies designed to strengthen the immune system and outsmart the disease. This is why
#CancerResearch
is so imperative.
#NationalCancerResearchMonth
1
6
33
In an interview with
@UroDocAsh
for
@UroToday
at
#ASCOGU
,
@UroCancerMD
discussed the results of our QUILT 3.032 clinical trial for BCG unresponsive NMIBC.
Read more here:
#BladderCancer
$IBRX
3
7
35
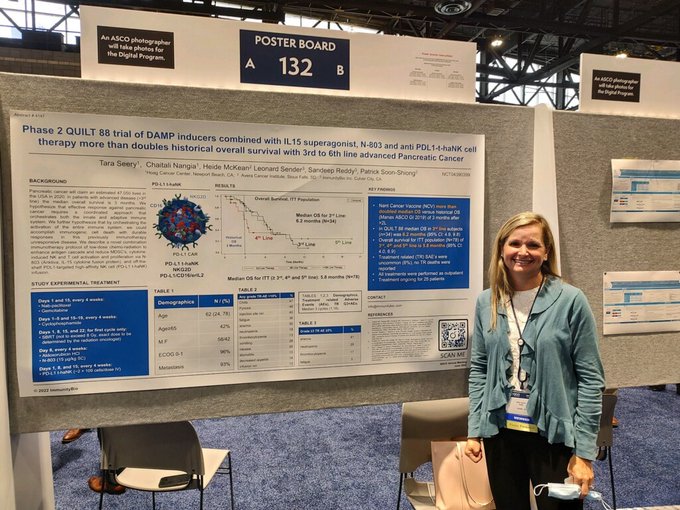
At
#ASCO22
, Dr. Tara Seery presents: Phase 2 clinical trial of DAMP inducers combined with IL15 superagonist, N-803, and anti–PD-L1 NK cell therapy more than doubles historical overall survival in patients with 3rd-6th line advanced pancreatic cancer.
1
9
32
In an interview with
@hannah_herner
for
@NashvillePost
, Dr. Sam Chang of
@VUMChealth
discusses latest data from
#bladdercancer
trial QUILT 3.032 and how it fills an unmet need for an immunotherapy that helps to fight the cancer.
@UroCancerMD
5
15
34
Bladder Trial: Data presented today at the AUA Meeting shows 72% of patients in a Phase 2/3 trial for BCG-unresponsive NMIBC have durable complete responses to treatment (median 19.9 months) and 85% of patients avoid cystectomy. Click for Webcast.
#IBRX
7
8
32
Our experimental therapy developed by
@DrPatSoonShiong
utilizes multiple parts of the immune system to destroy tumors - and it ultimately allowed both Senator Reid and Alex Trebek to prolong their lives.
Learn more:
0
8
34
We are leading the effort on next generation
#COVID
vaccines that are durable, accessible and effective against all future variants. Today, at the
@WhiteHouse
summit on
#COVID19
we discussed new opportunities to advance clinical research & bring innovative vaccines to market.
1
6
29
Dr. Sam Chang
@VUMedicine
will present the latest data from our Phase 2/3 bladder cancer trial at
#GU22
. Results surpass historical CR rates for FDA-approved therapies for BCG-unresponsive bladder cancer&exceed the AUA-FDA’s benchmarks for levels of complete remission&duration.
0
9
31
Existing COVID-19 vaccines assist in creating antibodies to the spike protein on the surface of the virus, while the oral T-cell vaccine we're working to develop targets the globe of the virus.
Learn more:
#COVIDVaccine
#ImmunityBio
#VaccineTrial
$IBRX
1
10
31
Globally there are an estimated 38M people living with HIV. Today, with
@umnmedschool
, we announced data from a Phase 1 “HIV Cure” Study which showed Anktiva stimulates latent HIV replication in CD4+ cells and increases immune cell activation. Read more:
0
9
32
Day 2 of
#AUA22
! Visit booth
#559
to find out how ImmunityBio is trying to prevent radical cystectomies for patients with BCG-unresponsive non-muscle invasive
#BladderCancer
1
7
30
#ICYMI
:
@UrologyPractice
published new findings from our QUILT 3.032 study on N-803 plus BCG in
#NMIBC
. Those who had a complete response to the combo therapy reported better physical function than those who did not achieve CR at 6 months. More here:
2
7
32
#ICYMI
: In an interview with
@NashvillePost
, Dr. Sam Chang,
@UroCancerMD
, of
@VUMChealth
discussed latest data from
#BladderCancer
trial QUILT 3.032 and how it fills an unmet need for an immunotherapy that helps to fight the cancer.
#AUA22
1
7
28
ImmunityBio is continuously pursuing new immunotherapies designed to attack disease by enhancing the patient’s immune system, not weakening it.
Read more about us here:
#clinicaltrials
#medicalresearch
#immunotherapy
#biotech
1
5
29
ImmunityBio has completed the acquisition of leasehold interest in a NY biotech manufacturing facility from
@AthenexInc
. The full-scale facility enables us to expand capabilities for large-scale manufacture of
#vaccine
and
#immunotherapy
candidates. More:
1
10
30
Together with
@theNCI
, we've opened a new
#clinicaltrial
studying our Tri-Ad5
#cancer
vaccines plus N-803 to prevent cancer in people with
#LynchSyndrome
. People with this syndrome have an increased risk for multiple cancers including
#ColonCancer
.
$IBRX
1
5
29
Recently,
@DrPatSoonShiong
spoke with
@corinnepurtill
to discuss our multivariant
#COVID19
vaccine candidate and clinical trials in South Africa. Read more about these types of vaccines via the
@LATimes
:
1
10
30
We’re looking forward to participating in the
#JefferiesHealthcare
conference on Friday at 8:15 am ET. We will be discussing our therapeutic portfolio and clinical progress across a range of highly challenging cancer types including bladder, pancreatic, and lung.
$IBRX
3
4
27
In an interview with
@biospace
@GeoffHerman1
, ImmunityBio founder
@DrPatSoonShiong
discusses our recent BLA submission for N-803 in BCG-unresponsive NMIBC & its potential to prevent cystectomies.
#bladdercancer
$IBRX
0
4
27
We are thrilled to invest in the city of Dunkirk and create hundreds of jobs for the community. Our goal is to manufacture vaccines for
#COVID
,
#HIV
and
#Cancer
. Thank you
@ChautauquaIDA
for sharing this video. $IBRX
1
10
27
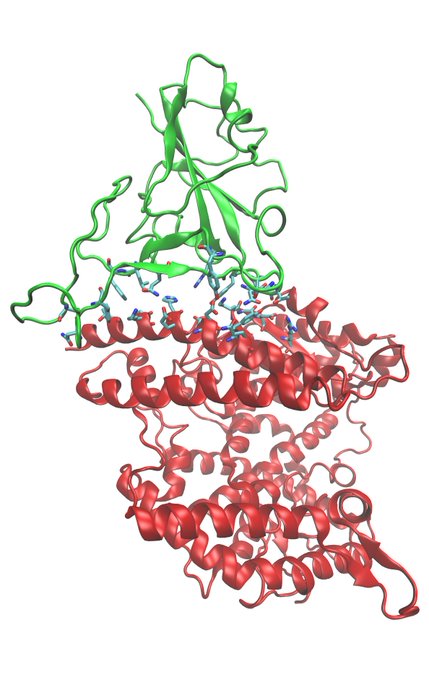
In clinical trials, the NANT Cancer Vaccine approach has been shown to induce complete remission across multiple tumor types in patients who previously progressed on standard-of care therapy.
#ImmunityBio
#NANTCancerVaccine
$IBRX
2
2
27
We are pleased to report that data from our bladder and pancreatic cancer clinical trials will be presented at ASCO in June.
#ASCO22
7
8
27
In a
@urotoday
interview, Dr. Alicia Morgans of
@DanaFarber
spoke to Dr.
@SWilliams_MD
with
@utmbhealth
about highlights from the non-muscle invasive
#bladdercancer
space, including the QUILT 3.032 study.
View the entire interview here:
#urology
#NMIBC
2
4
28
Our very own
@DrPatSoonShiong
delivered the annual David Packard Award Lecture at
@USUhealthsci
earlier today and focused on the future of
#cancer
treatments.
#CancerMoonshot
#Immunotherapy
4
5
26
We're pleased to announce the first participants have been enrolled in the
@lungmap
trial studying our novel potential treatment for Non-Small Cell Lung Cancer, a combination of Anktiva (N-803) + Keytruda. $IBRX Read more:
5
2
26
We are thrilled to partner with
@PresidencyZA
and others to expand our manufacturing capabilities in Africa to develop critically needed COVID-19 and cancer treatments. More here: $IBRX
0
0
23
“My concept of an ideal vaccine is one that doesn’t need refrigeration, offers quadruple immunity with antibody, T cell, mucosal and long-term memory protection, and most importantly, protects against the variants that are now emerging” – Patrick Soon-Shiong
#COVID19Vaccine
4
7
24
From the
@NEJM
: the mu variant shows a pronounced resistance to antibodies elicited by natural SARS-CoV-2 infection and by the BNT162b2 mRNA [Pfizer] vaccine. Next gen COVID-19 vaccines will be critical to minimizing the impact of mu and future variants. $IBRX
We need 2nd generation
@covid
vaccines now!
The next variant is concerning. NEJM letter just published!
12
16
59
2
8
25
Currently, patients with BCG-unresponsive
#bladdercancer
have very few options. Dr. Karim Chamie
@UCLAHealth
spoke with
@oncologytube
about the safety and efficacy of ImmunityBio’s QUILT 3.032 trial. More here:
2
7
23
Our Chief Medical Officer, Bobby Reddy, M.D., recently spoke with
@BioProcessOL
@mattpillar
about ImmunityBio’s plans to change the current standard of care to treat
#NMIBC
by applying N-803 to enhance the function of
#NKcells
. Listen here:
1
6
26
Alex Trebek, host of the long-running game show
#Jeopardy
, passed away 3 years ago today due complications related to
#pancreaticcancer
.
We're committed to finding better treatments w/ better outcomes for patients:
#PancreaticCancerAwarenessMonth
3
3
26
ImmunityBio’s Dr. Patrick Soon-Shiong talked earlier today with the BBC about second-generation
#COVID19
vaccines and the need to reduce both death and infection.
1
2
25
Approximately 38 million people worldwide live with
#HIV
, the virus that causes AIDS. ImmunityBio takes
#Pride
in our work to eliminate the disease and with it one of the world’s most serious public health challenges.
#immunotherapy
#HIVresearch
#AIDS
#awareness
#incluisivity
2
7
25
#DYK
#NMIBC
CIS affects tens of thousands of people every year. This week ImmunityBio submitted a Biologics License Application (BLA) for N-803 for intravesical administration plus
#BCG
for patients with BCG-unresponsive NMIBC CIS.
From
@YahooFinance
:
3
4
21
With big goals comes big growth! ImmunityBio grew by 243% last year when we merged with NantKwest. With our new facility in Dunkirk, the team will continue to grow. See our feature in
@businessinsider
's list of 17 fastest-growing drug companies of 2021:
6
3
23
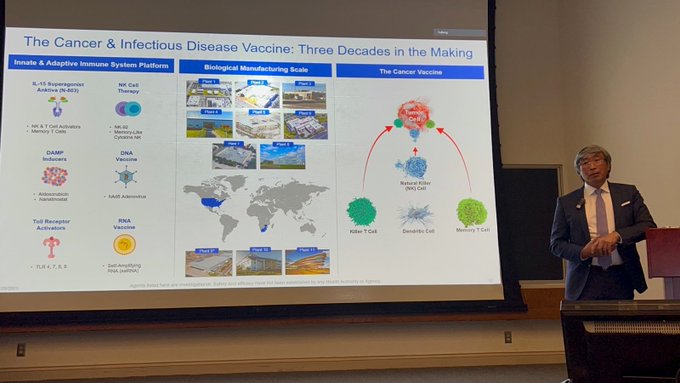
NK cell-based
#immunotherapies
are key to next-generation
#cancer
treatments.
The NK-92 cell became a key surrogate cell line used by researchers around the world and is the basis for our pioneering cell therapy platforms. Learn more:
$IBRX
#clinicaltrials
3
4
22
At ImmunityBio,
#diseaseawareness
is at the forefront of our mission.
Our
#immunotherapies
for treating
#cancer
and infectious diseases are currently being studied in a range of
#clinicaltrials
at various stages.
Learn more about our research:
$IBRX
1
3
25
From
@WhiteHouse
summit: Three strategies for next generation
#COVID
vaccines: target more than just an S target, engage T cell responses, combine adjuvants with protein vaccines. Tune in:
#COVID
0
4
24
At this year’s
#JefferiesHealthcare
conference, we look forward to providing a recap of the QUILT data shared earlier this week at ASCO and discussing the important role NK and T-cell activation play in fighting difficult-to-treat cancers.
$IBRX
#cancerresearch
#ASCO2022
5
4
23
Supporting the incredibly important work the Bladder Cancer Advocacy Network does on behalf of
#bladdercancer
patients and caregivers!
#bladderwalk
@BladderCancerUS
12
5
24
Our founder,
@DrPatSoonShiong
's, recent testimony to the U.S. Subcommittee on Africa, Global Health, and Global Human Rights addressed the need for a more durable and broad-acting
#COVID
vaccine that is room-temp stable to increase vaccinations in Africa.
2
6
23
The latest data from our QUILT 3.032 trial of N-803 plus BCG for adults with BCG-unresponsive
#NMIBC
CIS was published today in
@NEJMEvidence
. Read more about the results, which bring us closer to a potential treatment.
#BladderCancer
$IBRX
Pts with BCG-unresponsive carcinoma in situ and papillary non–muscle-invasive
#BladderCancer
treated with BCG and a novel IL-15 superagonist achieved a complete response rate of 71% meeting the trial’s primary endpoint.
#NMIBC
#OncoTwitter
@UCLAHealth
7
22
66
0
5
24
In the U.S.,
#pancreaticcancer
is the 4th leading cause of cancer-related death and has one of the highest mortality rates of all major
#cancers
, taking nearly 50,000 lives every year. Read more about our research here:
$IBRX
#Clinicaltrials
0
6
21
#Bladdercancer
is one of the most common cancers in men. Dr. Irv Cherny spoke with
@WMCActionNews5
, alongside his physician Dr. Sam Chang
@urocancerMD
, about how ImmunityBio's QUILT 3.032 trial prevented him from having a cystectomy after his diagnosis.
3
7
24