Skyler
@XC_Crypto
Followers
52
Following
157
Media
14
Statuses
70
Joined October 2017
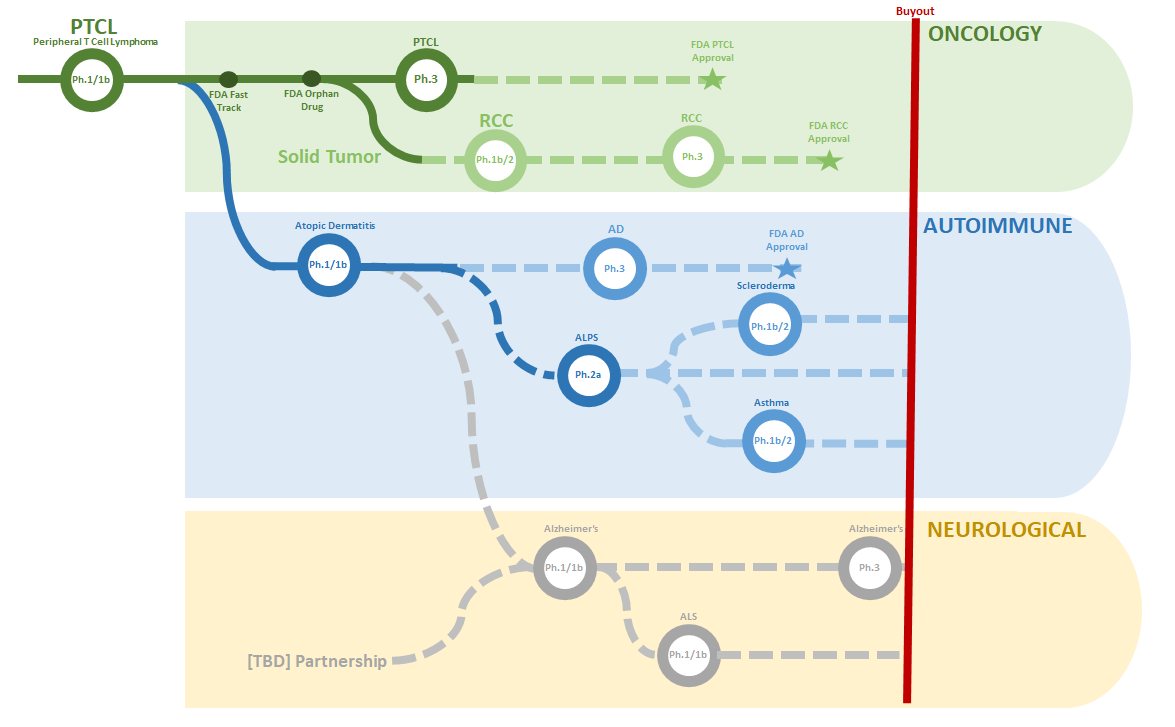
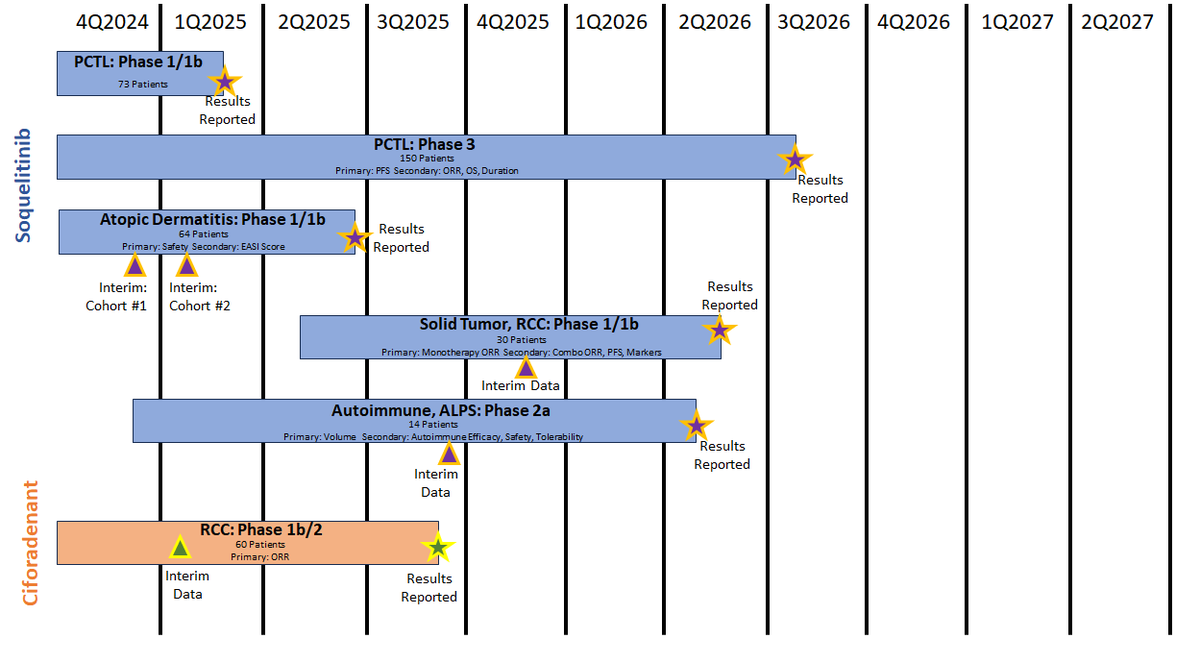
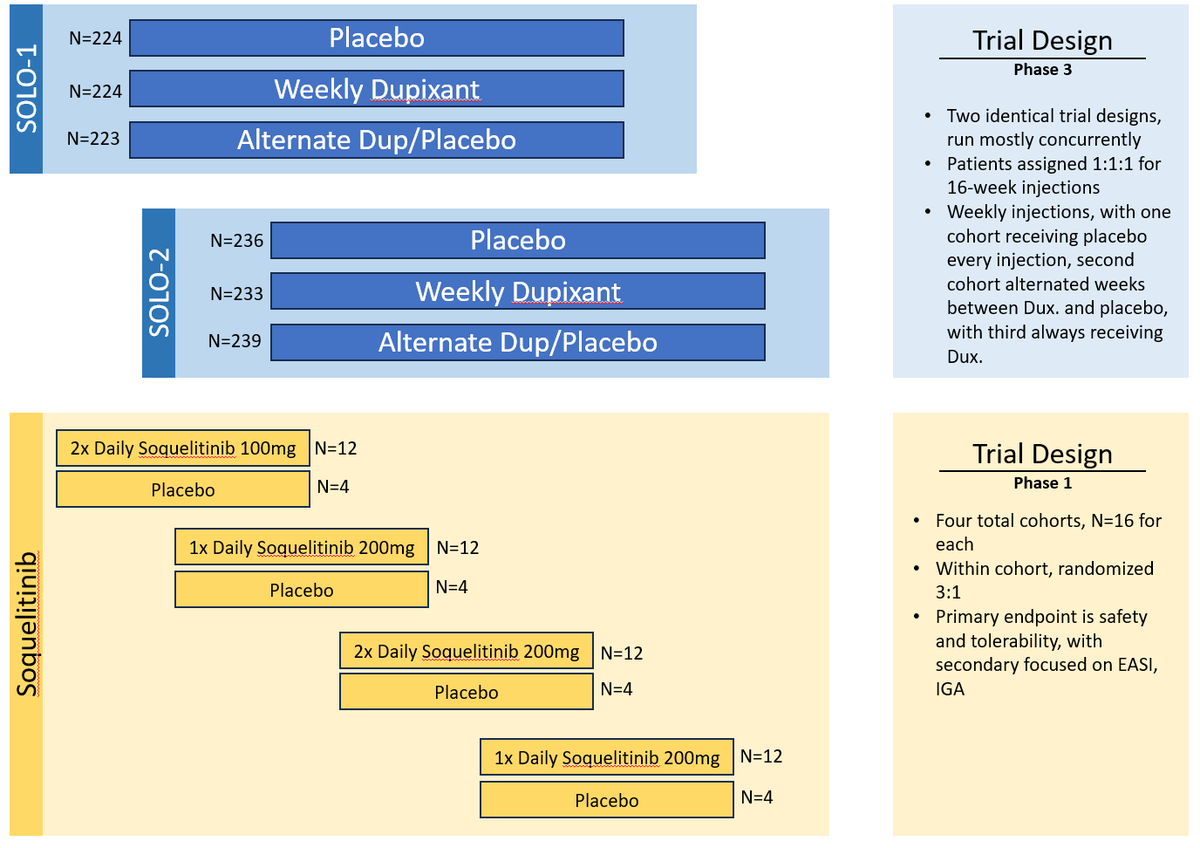
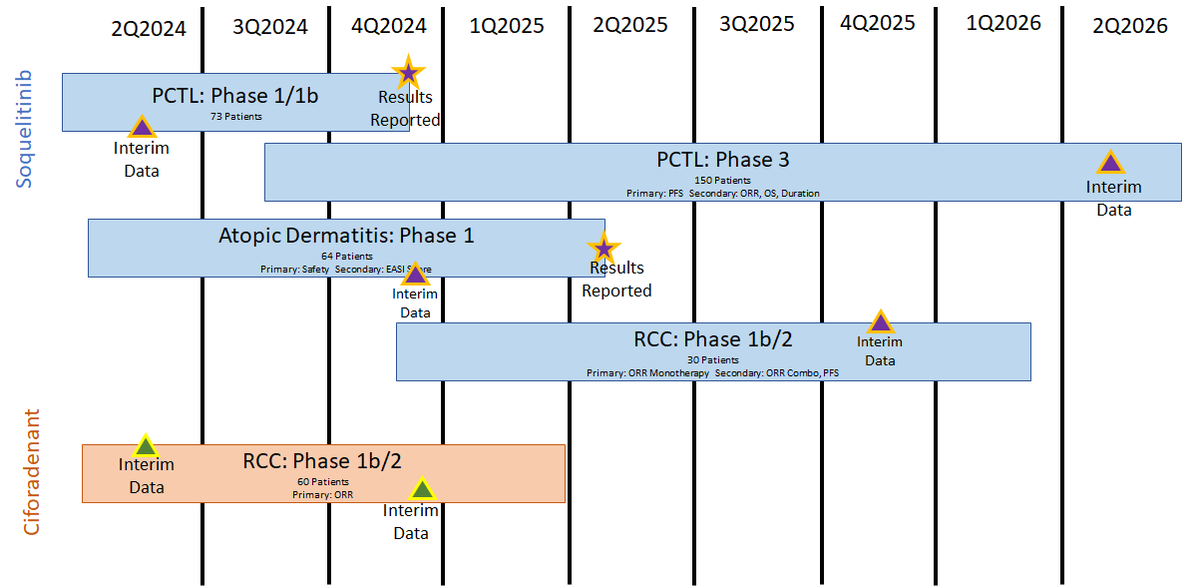
This can perhaps lead to larger multiples and spend less cash and time, to do it. When I look at all of the possible indications that $CRVS can use for #soquelitinib, I don't see them taking each one to Ph.3; just each with a simple n<50 trial with a placebo arm.
0
0
2
Shoutout to @ConradEnge98050 for all of his comprehensive analysis and status updates on $CRVS. It's certainly reinforced my investment thesis and helped to hammer home the science behind their MOA and pipeline. I'll owe you a steak dinner (or five) soon enough.
2
0
6
RT @Cryptochilly: @vincentbriatore #Neblio is massively undervalued right now with only 100m marketcap. Its competitors are all at 1 billi….
0
15
0